📌 3D Systems Clears the EU Bar and Enters Europe’s Denture Market
3D Systems has achieved a significant regulatory milestone, clearing the European Union’s stringent medical device requirements to enter the continent’s denture market. The company’s NextDent Jet Base, NextDent Jet Teeth, and the NextDent 300 MultiJet printer have all passed the Class IIa threshold under Europe’s MDR 2017/745 framework, the region’s governing standard for moderate-risk medical devices.
3D Systems已扫清欧盟壁垒,正式进军欧洲义齿市场
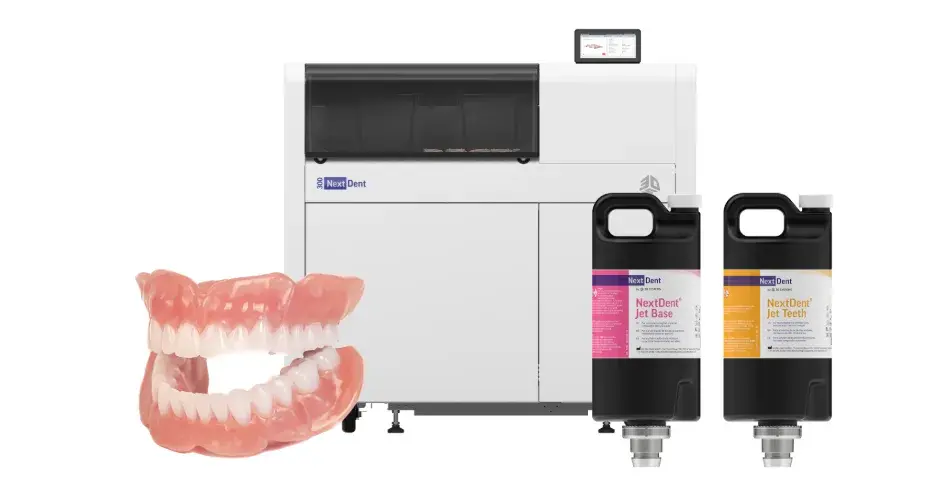
The practical effect is immediate: the full denture printing system can now be sold commercially throughout the European Union. Product availability starts May 4, two months ahead of what had been a summer target. This outcome extends a certification push that began in earnest when the company obtained broad-scope EU MDR recognition back in March 2026.
3D Systems在监管方面取得了重大里程碑,通过了欧盟严格的医疗器械要求,得以进入欧洲大陆的义齿市场。该公司的NextDent Jet Base、NextDent Jet Teeth以及NextDent 300 MultiJet打印机均已达到欧洲MDR 2017/745框架下的Class IIa门槛,该框架是欧洲针对中等风险医疗器械的监管标准。
The combination of the NextDent 300 hardware with the Jet Base and Jet Teeth materials allows dental labs to go from digital file to finished, patient-fitted denture in one print run—no secondary curing required afterward. The result is a monolithic, multi-material jetted denture, structurally unified and ready for finishing straight out of the printer. Speed, dimensional precision, and repeatability all improve over what conventional handcrafted methods can deliver.
实际效果立竿见影:全口义齿打印系统现可在整个欧盟范围内进行商业销售。产品将于5月4日起售,比原定夏季目标提前了两个月。这一成果延续了该公司自2026年3月获得广泛范围的欧盟MDR认证以来持续推进的认证工作。
The company frames this as the completion of its dental portfolio, covering the full patient treatment spectrum from alignment correction and bite protection through to restoration and full tooth replacement.
NextDent 300硬件与Jet Base和Jet Teeth材料的组合,使牙科实验室能够从数字文件到完成患者适配的义齿一次打印成型——无需后续二次固化。最终得到的是一个整体式、多材料喷射成型的义齿,结构统一,出机即可进行精加工。在速度、尺寸精度和可重复性方面,均优于传统手工制作方法。
“Achieving Class IIa MDR certification for our NextDent Jetted Denture Solution is a major milestone that validates the strength of our clinical data, quality systems, and innovation pipeline. By launching in Europe on May 4, well ahead of our original summer timeline, we are immediately expanding access to this transformative technology for dental labs and clinics across the region. This clearance substantially increases our available addressable market for this high-value platform. As adoption accelerates in both the U.S. and Europe, we expect it to drive significant recurring revenue through premium materials while reinforcing our position as the leading full-service provider in digital dentistry,” said Jeffrey Graves, Ph.D., President and CEO.
该公司将此视为其牙科产品组合的完善,覆盖了从矫正对齐、咬合保护到修复及全口牙齿替换的完整患者治疗范围。
For 3D Systems, regulatory approval is not a formality that follows product development—it is the market entry strategy itself. The company has been systematically building its certification infrastructure across the EU, treating MDR compliance as the foundation on which commercial scale is constructed. In an interview, Chuck Stapleton, former VP and General Manager for Dental at 3D Systems, noted that MDR requirements in the EU are significantly slowing down the pace at which products can be brought to market, which means whoever clears that bar first effectively locks out slower-moving rivals.
“我们的NextDent Jetted Denture Solution获得Class IIa MDR认证是一个重要里程碑,验证了我们临床数据、质量体系和创新管线的实力。通过于5月4日在欧洲上市,远早于我们原定的夏季时间表,我们正在立即扩大该地区牙科实验室和诊所获取这项变革性技术的渠道。此次获批显著增加了我们这一高价值平台的可寻址市场。随着在美国和欧洲的采用率加速增长,我们预计其将通过优质材料推动可观的经常性收入,同时巩固我们作为数字牙科领域领先全服务提供商的地位,”总裁兼首席执行官Jeffrey Graves博士表示。
The same logic is playing out across the competitive landscape. Stratasys recently secured CE Class IIa certification for its TrueDent resin portfolio, making it the first high-aesthetic, single-material 3D printed denture system to earn that designation in Europe—a direct signal that major players now treat regulatory clearance as the primary competitive move, not the product launch itself.
对于3D Systems而言,监管批准并非产品开发后的例行程序——它本身就是市场进入策略。该公司一直在系统性地建设其在整个欧盟的认证基础设施,将MDR合规视为构建商业规模的基础。在接受采访时,3D Systems前牙科业务副总裁兼总经理Chuck Stapleton指出,欧盟的MDR要求显著减缓了产品上市速度,这意味着谁先通过这一门槛,谁就能有效阻止行动较慢的竞争对手。
Even smaller operators have followed the same path: Dentafab obtained CE Class IIa certification for its PowerResins Temp and Denture materials, explicitly framing the accreditation as the mechanism to increase the spread of its products across European countries. Across company sizes, the pattern is consistent: in regulated medical markets, the approval is the product.
同样的逻辑正在整个竞争格局中上演。Stratasys近期为其TrueDent树脂产品组合获得了CE Class IIa认证,使其成为欧洲首个获得该认证的高美观度、单材料3D打印义齿系统——这直接表明,主要玩家现在将监管批准视为首要竞争举措,而非产品发布本身。
For dental labs and clinics across Europe, this certification wave means faster access to advanced digital denture solutions that deliver consistent, high-quality results. The ability to print complete dentures in a single run without post-curing dramatically streamlines workflows and reduces turnaround times. As more companies achieve regulatory clearance, the adoption of digital dentistry will accelerate, offering patients better-fitting, more durable dentures produced with precision that traditional methods cannot match.
即使是较小的运营商也遵循了相同的路径:Dentafab为其PowerResins Temp和Denture材料获得了CE Class IIa认证,明确将该认证视为扩大其产品在欧洲各国覆盖范围的机制。无论公司规模大小,模式是一致的:在受监管的医疗市场中,批准即是产品。
If you are working with 3D printing models for dental applications, staying informed about certified materials and systems is essential for compliance and quality. Explore our collection of premium STL files designed for professional dental labs and clinics.
对于欧洲各地的牙科实验室和诊所而言,这股认证浪潮意味着更快的获取
Looking for high-quality STL files? Browse our collection at 3dmis.com!
Original source: View Original
🚀 开启你的下一个 3D 打印项目
想要高质量的 STL 文件?浏览我们丰富的优质 3D 模型库,从流行文化角色到桌游 mini,应有尽有——尽在 3dmis.com!
🚀 Start Your Next 3D Printing Project
Looking for high-quality STL files? Browse our vast collection of premium 3D models at 3dmis.com!
📌 编者按:本文改编自行业最新资讯。查看原文

